INN & Regulatory Reference
NexCAR19™
A New Hope for Hard to Treat B-Cell Cancers
An Overview of NexCAR19™
A breakthrough in precision oncology, NexCAR19™ (Talicabtagene autoleucel) is a second-generation, autologous, CD19-directed chimeric antigen receptor (CAR) T-cell immunotherapy. Built on a fully humanized molecular platform, it is precision-engineered to address the critical unmet need in relapsed or refractory B-cell malignancies.
NexCAR19™ is a prescription therapy indicated for specific relapsed or refractory B-cell Non-Hodgkin’s Lymphomas and B-cell Acute Lymphoblastic Leukaemia in patients whose frontline or other standard treatments have been unsuccessful.
CAR-T cell therapy
Response Rate
Response Rate
Longest Remission
Success Rate
Turnaround
NexCAR19™ (Talicabtagene autoleucel) - Approved Indications
Relapsed/Refractory B-cell Non-Hodgkin Lymphoma (R/R B-NHL)
After 1 or more lines of prior systemic therapy · Age 15 years and above
- ✓ Diffuse Large B-cell Lymphoma (DLBCL)
- ✓ Primary Mediastinal B-cell Lymphoma (PMBCL)
- ✓ Follicular Lymphoma (FL)
- ✓ Mantle Cell Lymphoma (MCL)
- ✓ Marginal Zone Lymphoma (MZL)
- ✓ High-Grade B-cell Lymphoma (HGBL)
~40% of DLBCL patients relapse after first-line therapy; >60% are ineligible for autologous stem cell transplantation (ASCT), representing a significant unmet need addressed by NexCAR19™.
Relapsed/Refractory B-cell Acute Lymphoblastic Leukemia (R/R B-ALL)
After 1 or more lines of prior therapy · Age 15 years and above
- ✓ CD19-positive B-cell precursor ALL confirmed by flow cytometry or immunohistochemistry
- ✓ Relapsed or refractory after ≥1 prior line of therapy, including one intensive chemotherapy regimen
- ✓ Applicable to adolescents (age 15–18) and adults - broadening access vs. paediatric-only CAR-T therapies
- ✓ No active CNS involvement at time of treatment (CNS-1 or CNS-2 only after clearance)
Historical B-ALL survival after relapse remains poor at ~41% with conventional chemotherapy. NexCAR19™ provides a curative-intent option where allogeneic SCT may not be feasible or accessible.
Unmet Needs in Relapsed/Refractory B-Cell Malignancies
Transforming outcomes for patients with relapsed/refractory B-cell malignancies – where current treatments fall short. Despite significant advances in oncology, a large proportion of patients relapse after frontline therapy, and for many, curative options remain out of reach. These numbers reflect the scale of the challenge and the urgent need for more effective, accessible treatments.
More than 550,000 new B-cell non-Hodgkin lymphoma cases and over 100,000 B-ALL cases are diagnosed globally each year - representing one of the largest unmet burdens in oncology.
Source: GLOBOCAN 2022; PMC11829893Up to 40% of DLBCL patients and 20-25% of ALL patients develop relapsed or refractory disease after frontline therapy - with minimal durable responses to standard salvage regimens.
Source: ASCO EDBK_390802; Tandfonline 2024More than 60% of patients with relapsed DLBCL are not eligible for stem cell transplant due to age, comorbidities, or chemo-refractory disease. Median survival with conventional therapy is only ~6 months.
Source: Semanticscholar; PubMed 30213390India reports ~50,000 new leukemia and ~40,000 NHL cases annually. Only 0.09% of India's population is registered as potential stem cell donors - a fraction of rates in Western countries.
Source: GLOBOCAN 2022 India; PMC11893321Patient Journey - HCP Workflow
A coordinated 5-step process from eligibility confirmation to infusion and monitoring
 Important: This workflow represents a generalized treatment pathway. Patient-specific variations in sequencing, timelines, and interventions are expected and are determined by the treating oncologist/haematologist based on disease progression, clinical status, and institutional protocols.
Important: This workflow represents a generalized treatment pathway. Patient-specific variations in sequencing, timelines, and interventions are expected and are determined by the treating oncologist/haematologist based on disease progression, clinical status, and institutional protocols.Recommended Patient Eligibility Criteria
Assess all criteria before initiating the NexCAR19 treatment process.
General Eligibility Criteria
- ECOG Performance Status: 0–2
- Ejection Fraction ≥45% (2D Echo, within 6 weeks)
- Peripheral Blood ALC ≥500/μL or Absolute CD3+ T cell count ≥150/μL
- AST/ALT ≤3× ULN | Total Bilirubin ≤2× ULN
- Creatinine Clearance ≥30 mL/min
- Well-preserved lung function
- Well-controlled pre-existing comorbidities
- Negative viral markers (HIV, HBV, HCV, CMV)
- CNS disease well-mitigated if involved
- Absence of active infection or acute inflammation
- Active uncontrolled infections or acute inflammation
- Uncontrolled or severe cardiac conditions
- Pregnancy or breastfeeding (contraindicated)
Disease-Specific Criteria
- After 1 or more prior lines of therapy
- Age 15 years and above
- Relapsed or refractory after standard lines of therapy
- CD19-positive confirmed by flow cytometry or IHC
- Measurable disease per Lugano criteria at screening
- Prior ASCT patients may be eligible — evaluate case-by-case
- Leukapheresis feasibility and timing confirmed
- No active CNS lymphoma at time of treatment
Patient Journey Overview
Step 1: Eligibility Assessment
Determine Suitability for NexCAR19 Therapy
Evaluate the patient against NexCAR19’s eligibility criteria, including diagnosis, disease status, prior lines of therapy, and performance status.
Step 2: Slot Scheduling
Coordinate Leukapheresis Timing Based on Clinical Readiness
Once eligibility is confirmed, our team will contact you to schedule a leukapheresis slot at your facility or designated center.
Please ensure:
-
Patient is clinically stable and fit for leukapheresis
-
Appropriate drug washout periods are observed
-
Any bridging therapy is planned with manufacturing timelines in mind
Step 3: LSM Collection
Timely & Controlled Pickup of Leukapheresis Material
Upon successful leukapheresis:
-
ImmunoACT or its certified logistics partner will coordinate fresh refrigerated pickup of the Leukapheresis Starting Material (LSM)
-
Ensuring correct labeling, documentation, and chain-of-custody protocols are followed
Step 4: Manufacturing
Stay Informed at Key Checkpoints During Manufacturing
You will receive notifications at the following stages:
-
LSM acceptance and initiation of manufacturing
-
Completion of in-process and final quality checks
-
Expected product readiness and dispatch timeline
Step 5: Infusion & Monitoring
Product Delivery, Infusion Scheduling, and Post-Infusion Care
NexCAR19 will be delivered in advance of your selected infusion window:
-
Confirm patient’s fitness for lymphodepletion and infusion
-
Administer NexCAR19 as per prescribing information
-
Follow structured monitoring protocol post-infusion, including CRS/ICANS management if needed
What are the Side Effects of NexCAR19?
NexCAR19™ may cause side effects that are severe and/or life-threatening.








How does NexCAR19™ work - Scientific Profile
A humanized CD19-targeting CAR-T construct with optimized persistence and reduced immunogenicity
-
Humanized scFv — CD19 TargetingA humanized single-chain variable fragment (scFv) antibody specifically targets CD19 markers on B-cells. Humanized design significantly reduces immunogenicity compared to murine-based CAR constructs.
-
Optimized Flexible Spacer (CD8 Hinge)An optimized flexible spacer enhances target binding efficiency and allows optimal orientation of the scFv for CD19 engagement.
-
4-1BB Co-stimulatory DomainSpecialized 4-1BB co-stimulatory signaling significantly boosts T-cell activation and promotes long-term T-cell persistence — contributing to durability of response beyond 24 months in select patients.
-
CD3ζ Signaling DomainCD3ζ signaling components trigger potent T-cell immune response upon CD19 binding, initiating cytotoxic killing of CD19+ malignant B-cells.
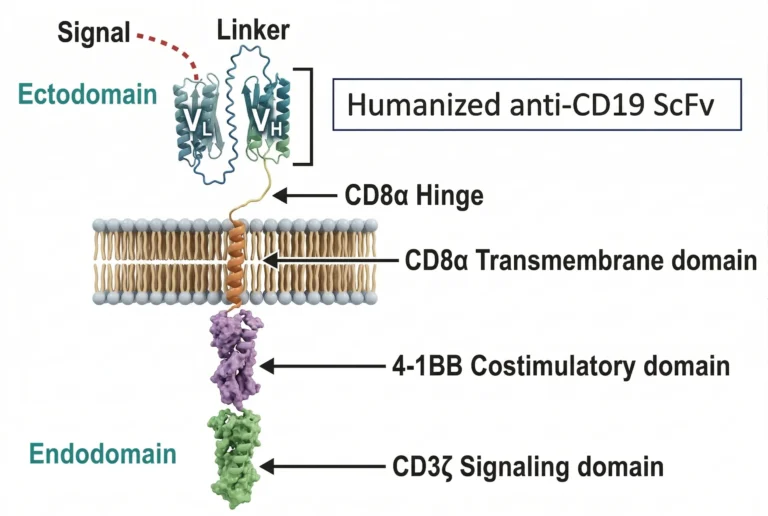
Proven Clinical Performance
Our anti-CD19 binding technology has demonstrated remarkable persistence in clinical settings, with documented durability of action extending beyond 24 months in select patients. The humanized ScFv component delivers exceptional efficacy while contributing to a reduced incidence of severe treatment-related toxicities.
Superior Therapeutic Profile
NexCAR19™ modified T-cells offer a comprehensive treatment solution:
- Safety: Rigorously tested cellular product with established safety profile
- Durability: Long-lasting anti-cancer activity for sustained disease control
- Effectiveness: Consistent response rates across diverse patient populations
Optimized Dosing Strategy
With an efficacy threshold of more than 5 million CAR-T cells per kilogram of body weight, NexCAR19™ achieves exceptional tumor penetration and maintains effectiveness even in challenging high-risk patient populations.
NexCAR19™ is a type of CAR-T cell therapy. T-cells are naturally made by your body as an advanced defense against viruses and cancer cells. As they mature, they develop specific connectors (receptors) to target key signals in cancer cells. However, cancers can escape the inbuilt defense mechanism of T-cells, which can lead to an increase in tumor burden and can result in the survival of cancer cells and a further increase in tumor burden. CD19 is a protein commonly present on the surfaces of certain B-cell cancers.
Our scientists have designed instructions for your T-cells to express unique proteins called Chimeric Antigen Receptors (CARs) on their surface, which will enable them to bind to a specific target on the cancer cells. These instructions are delivered genetically using a vehicle known as a lentiviral vector.NexCAR19 targets a marker called CD19, which is commonly present on the surface of cancerous B-cells.
1. Isolate
Select and activate T-cells from the patient’s blood sample.
2. Program
Deliver genetic instructions to T-cells using viral vectors.
3. Engineer
Enable T-cells to express Chimeric Antigen Receptors (CARs).
4. Expand
Multiply CAR-T cells to achieve the therapeutic dose.
5. Infuse
Administer CAR-T cells to the patient for targeted cancer treatment.
Smarter CAR-Construct & Design for Safer Treatment
At the heart of ImmunoACT’s innovation is a next-generation cell and gene therapy platform, purpose-built to deliver affordable efficacy without compromising safety. Our approach blends cutting-edge science with thoughtful design to make these therapies more accessible and tolerable.
Our CAR-T constructs use fully humanized components, making them more “familiar” to the immune system. This reduces the risk of rejection, minimizes off-target toxicity, and improves patient experience by lowering the need for intensive supportive care.
Each CAR we design includes:
- A single-chain variable fragment (scFv) tailored for precise tumor targeting
- A flexible hinge (spacer) to enable optimal antigen engagement
- A CD8α transmembrane domain for enhanced stability
- A 4-1BB co-stimulatory domain to support T-cell survival and sustained action
- A CD3ζ signaling domain to trigger strong and specific immune responses
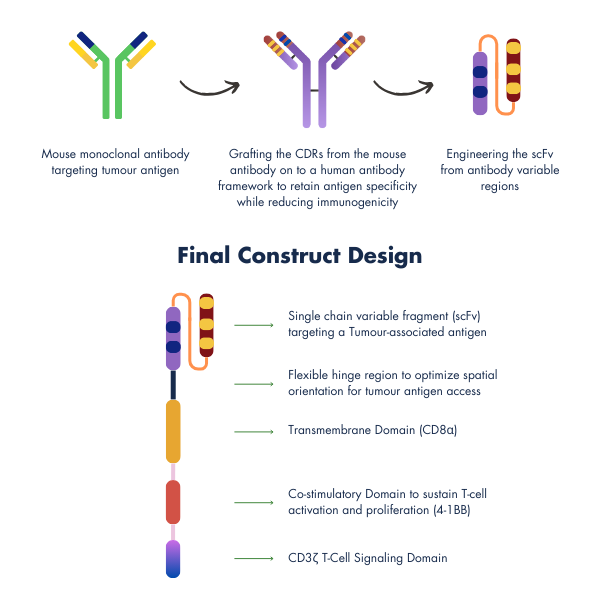
Our CAR-T therapies integrate humanized sequences and undergo rigorous optimization to ensure potency, persistence, and safety. By reducing the likelihood of severe toxicities, we are not just improving patient safety, we are redefining how cell therapies can be delivered.
The gene sequence of the CAR construct is first integrated into a plasmid, which is then used to produce a lentiviral vector. This vector delivers the CAR gene into T cells, enabling them to recognize and attack cancer cells.
NexCAR19™ Manufacturing Process
1. Leukapheresis
This forms the foundational starting material for autologous CAR-T cell manufacturing.
2. Monocyte Depletion
3. T-Cell Activation
4: Lentiviral Transduction
5: CAR-T Cell Production
6: Expansion to Target Dose
7: Formulation & Cryopreservation
Why NexCAR19™ ?
NexCAR19™ demonstrates strong clinical efficacy with rapid response rates, durable remissions, and a streamlined manufacturing timeline – addressing critical unmet needs in relapsed/refractory B-cell malignancies.
Safety Profile of NexCAR19™
NexCAR19™ demonstrates a predictable and manageable safety profile, characterised by low rates of severe immune-mediated toxicities and consistent outcomes across real-world settings.
Relapsed / Refractory B-ALL
Relapsed / Refractory B-NHL
Individual patient outcomes may vary.
The Hope Initiative - Clinical & Institutional Support
The Hope Initiative is designed to support not just patients – but the physicians and institutions who deliver care. It is an integrated operational and clinical framework built to enable safe, scalable, and outcome-driven CAR-T therapy across India.
From institutional onboarding and protocol alignment to real-time toxicity management support and long-term outcome capture – The Hope Initiative is built around your clinical needs.
Institutional Onboarding
Structured onboarding frameworks to establish site readiness, align clinical protocols with global standards, implement toxicity management frameworks, and build institutional capability for safe and efficient CAR-T delivery.
CAR-T Centre EnablementToxicity Management Support
Access to an experienced HCP helpline providing real-time clinical guidance to managing teams on post-infusion toxicity management - including CRS and ICANS - strengthening preparedness at every stage of post-CAR-T care.
On-demand Clinical GuidanceVein-to-Vein Coordination
End-to-end coordination across apheresis logistics, manufacturing timelines, and infusion scheduling — ensuring no critical moment is missed and your clinical team can stay focused on patient outcomes.
Seamless Operational SupportACT.360
A dedicated monitoring and data platform enabling long-term safety surveillance, real-world evidence capture, and structured outcome documentation - giving you and the broader clinical community actionable insights from every patient's journey.
Real-World EvidencePublications
Read our publications, published in prestigious journals such as The Lancet Haematology, Molecular Cancer Therapeutics by AACR and Blood by the American Society of Hematology.
Novel humanized CD19-CAR-T (Now talicabtagene autoleucel, Tali-cel™) cells in relapsed/ refractory pediatric B-acute lymphoblastic leukemia- an open-label single-arm phase-I/Ib study
Article
Apr 24, 2025Talicabtagene Autoleucel for Relapsed or Refractory B-cell Malignancies: Results from an Open-label, Multicentre, Phase 1/2 Study
Article
Apr 1, 2025Excellent Safety Profile of a Low-Cost Novel Humanized CD19 CAR T-Cell Therapy, Actalycabtagene Autoleucel : Potential Impact on Access and Feasibility
Abstract
Dec 9, 2023
NexCAR19™ Prescribing Information Guide
Essential prescribing information and medication guide for NexCAR19™ (Talicabtagene autoleucel), a CAR-T cell therapy for treating relapsed/refractory B-cell lymphomas and B-cell acute lymphoblastic leukemia. Includes detailed dosing instructions, safety information, and patient counseling guidelines.
Partnered Hospitals
Our strong association with over 130 + leading cancer treatment hospitals in India ensures hassle-free treatment with our CAR-T cell therapies.
